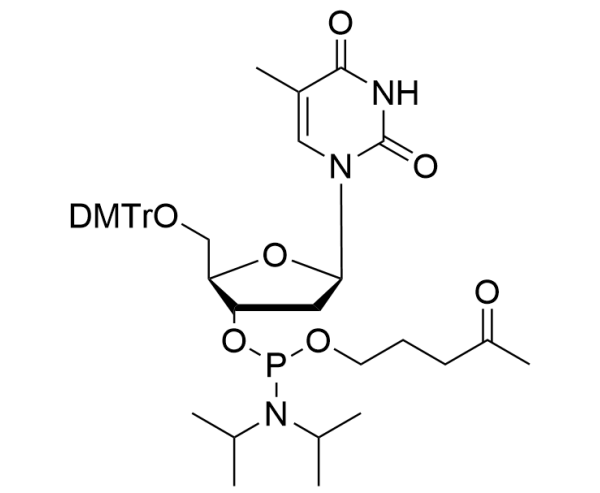
dT Oxopentyl Phosphoramidite
| Chemical name |
5’-O-(4,4’-Dimethoxytrityl)-3’-O-(N,N-diisopropylamino)[4- oxopentyl]phosphinyl-2’-deoxythymidine |
| CAS number | 370596-61-5 |
| Molecular formula | C42H54N3O9P |
| Molecular weight | 775.87 g/mol |
| Mass contribution | 390.3 g/mol |
| Storage Conditions | Keep in a dark and dry place at −20°C. Store under argon. |
| Usage | Prepare 0.067M solution of dT Oxopentyl Phosphoramidite in dry MeCN. Cap A solution must be the phenoxyacetyl anhydride version of the reagent, not acetic anhydride. Deblock in 0.05M K2CO3 in MeOH, room temp, 16-20 h. |
Oxypentyl-modified Nucleoside Phosphoramidites are designed for the synthesis of oligonucleotides containing a thermolabile 3'-blocking group, which prevents primer hybridization and extension at low temperatures and is automatically removed during high-temperature activation.
Oligonucleotides synthesized using these phosphoramidites function as built-in hot-start primers, eliminating premature annealing and nonspecific extension during reaction setup and thermal cycling.
Key features:
- Temporary 3′-blocking with thermal removal
- Suppression of nonspecific primer binding
- Reduction of dimer formation
- Compatibility with standard oligonucleotide synthesizers
Oxypentyl phosphoramidites provide a chemical approach to hot-start PCR, allowing precise control of primer activity without the use of modified enzymes.